Official Collaborated with:

Pipeline -
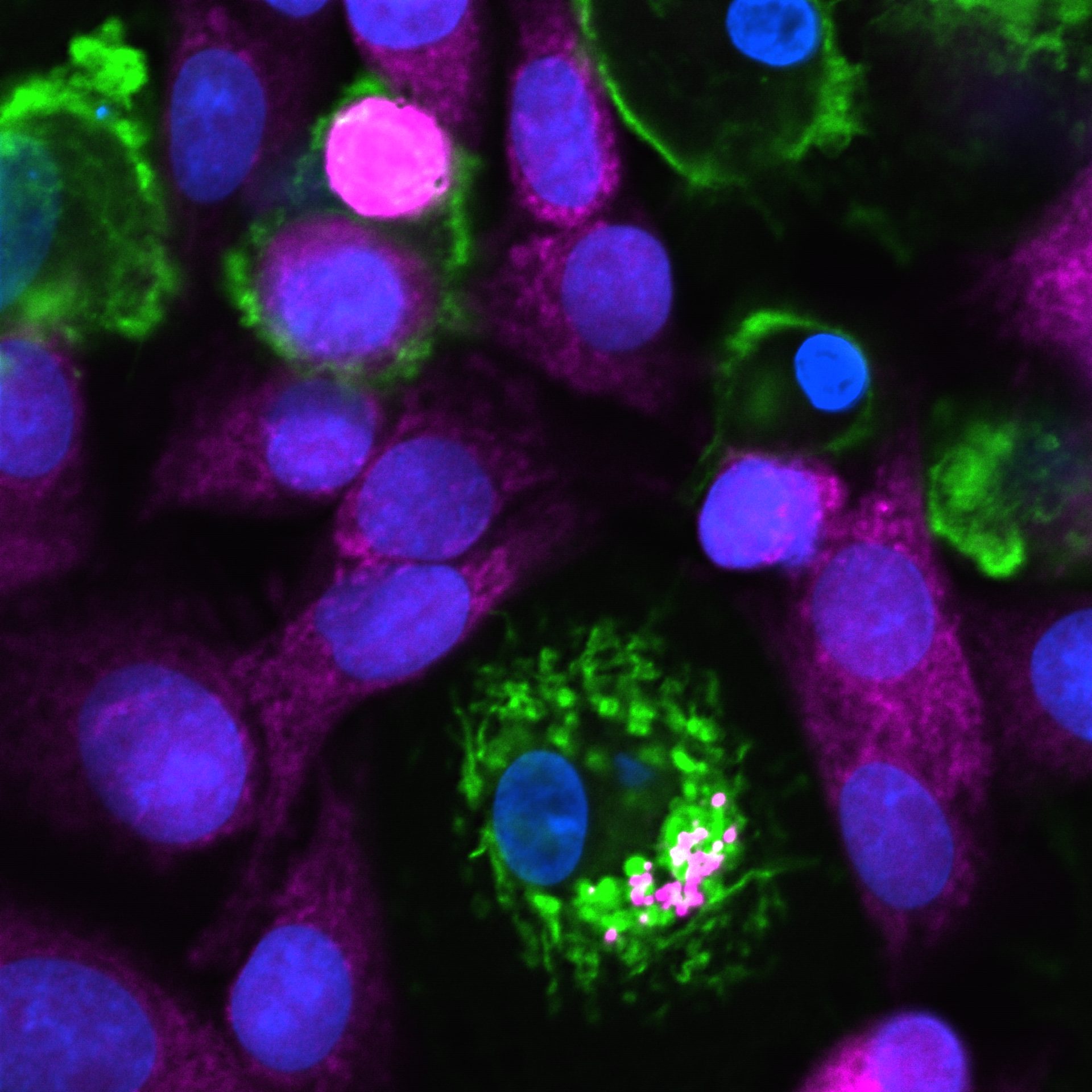
New Stem Cell Technology
Taking advantage of totipotency features of Expanded Potential Stem Cells (EPSCs) and unique properties to development novel animal cloning technologs, CTSCB maps cell lineage atlas from human EPSCs to cell teypes relevant to regenerative medicine and immunotherapies. The new human cell lineage knowledge will directly inform the development of optimised protocols for efficient generation of specific cell types from human EPSCs.
More Info
EPSCs for Regenerative Medicine & Human Disease Study
Clinical grade Mesenchymal Stem Cells (MSCs) can be differentiated from human Expanded Potential Stem Cells (EPSCs) which are easier in culture, genetically and epigenetically stable, and allow more efficient genome-editing to generate modified MSCs that provide better therapeutic potential and further reduce their immune rejection after transplantation.
More Info
EPSCs for Genomic Medicine of Immune Disease
CTSCB uses the Expanded Potential Stem Cells (EPSCs) technology to link genotype to phenotype through genetically defined stem cell-based models of human immune disease.
More Info

Learn more about our patented technology
Empowering the Future with Expanded Potential Stem Cells
We invent with EPSC Technology.
We invent with EPSC Technology.
Totipotency Features
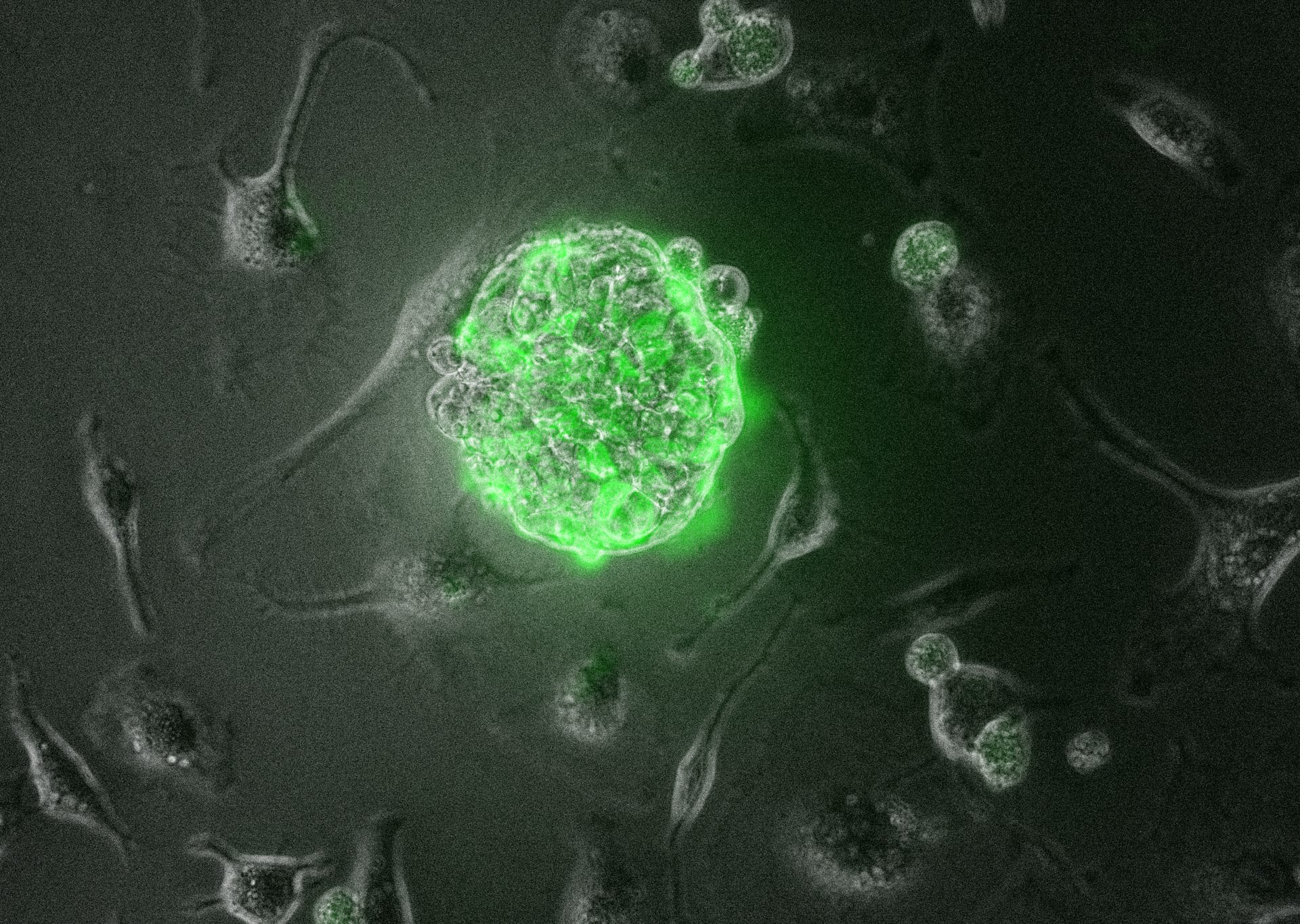
EPSCs closely resemble early human embryos at the 8-cell to morula stage.
Genetically Stable
EPSCs are easy to generate, genetically stable, and highly reproducible.
Precise Genome Editing
EPSCs offer efficient genome editing, enabling breakthrough in targeted disease models, target validation, and personalized therapy.
Personalized EPSCs
With a few drops of blood, we produce stem cells at scales large enough to benefit thousands.
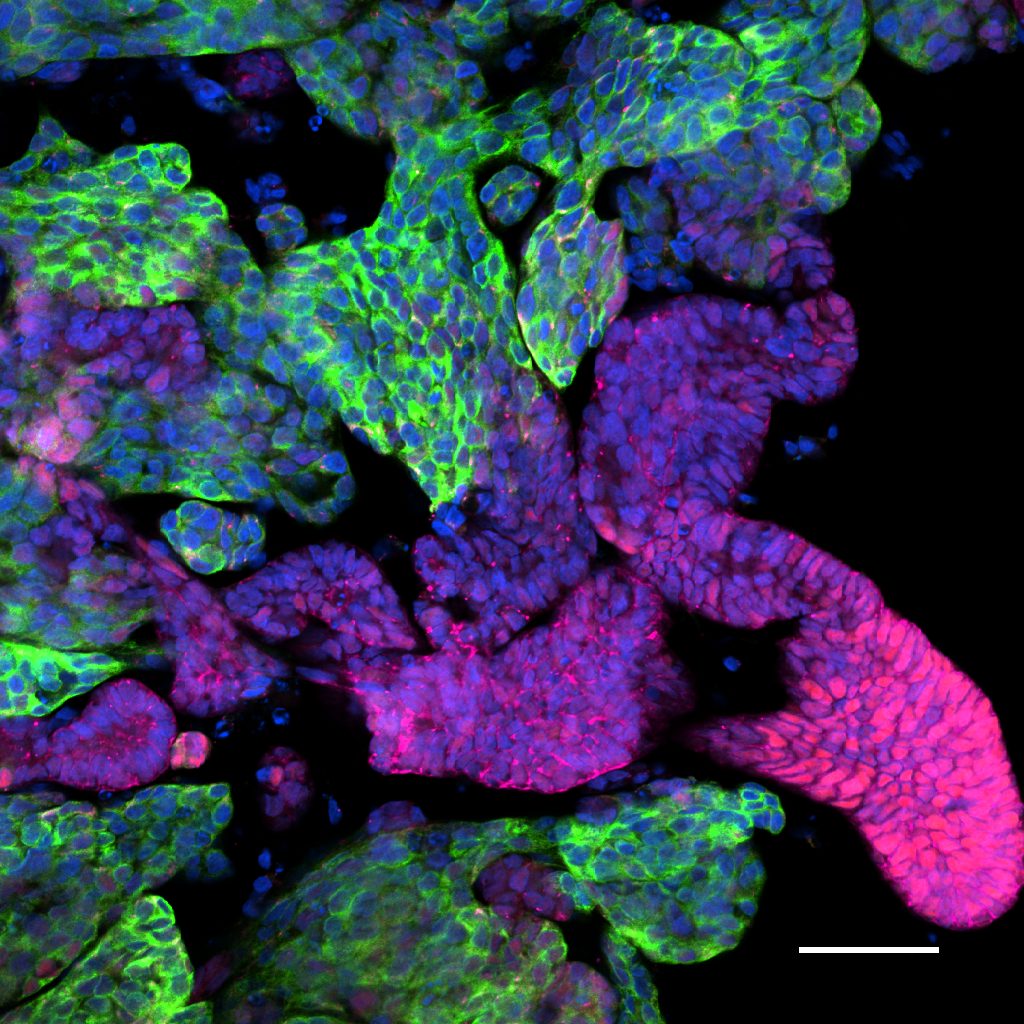
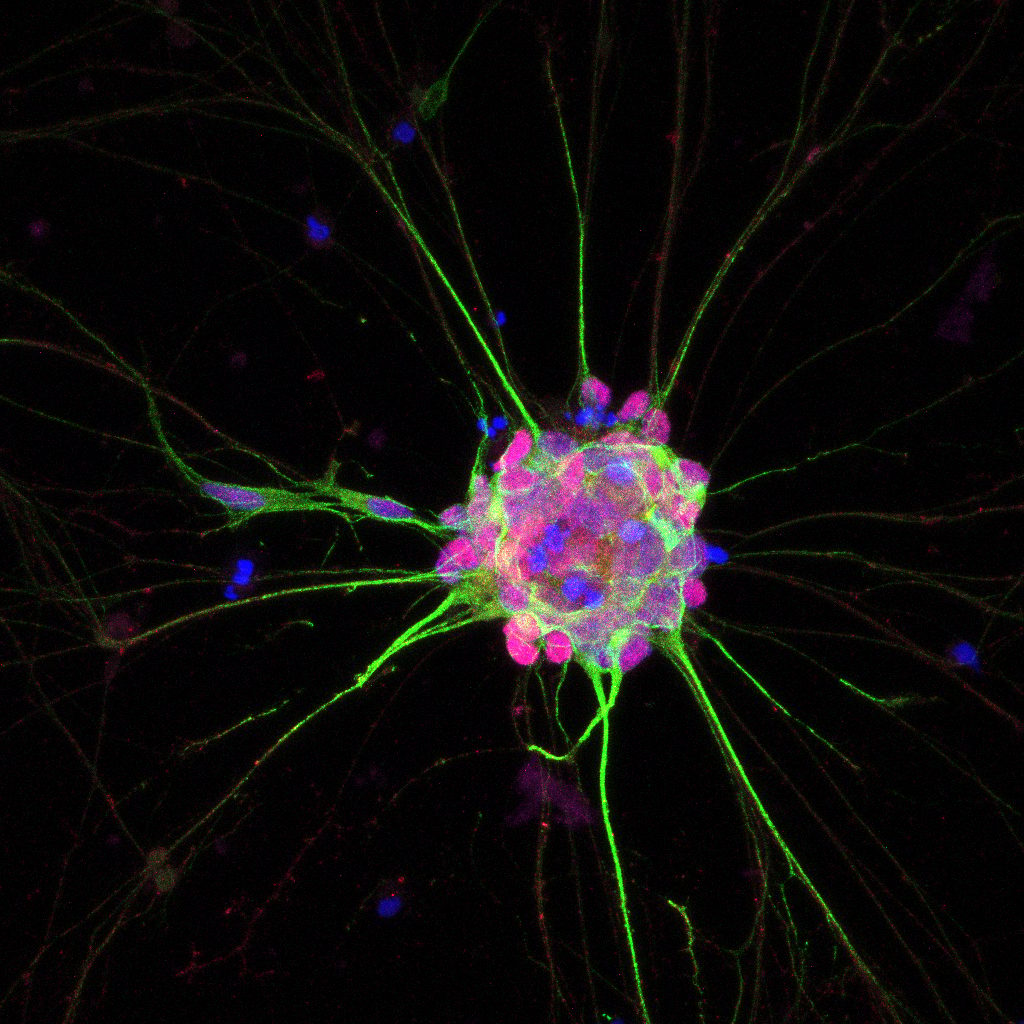
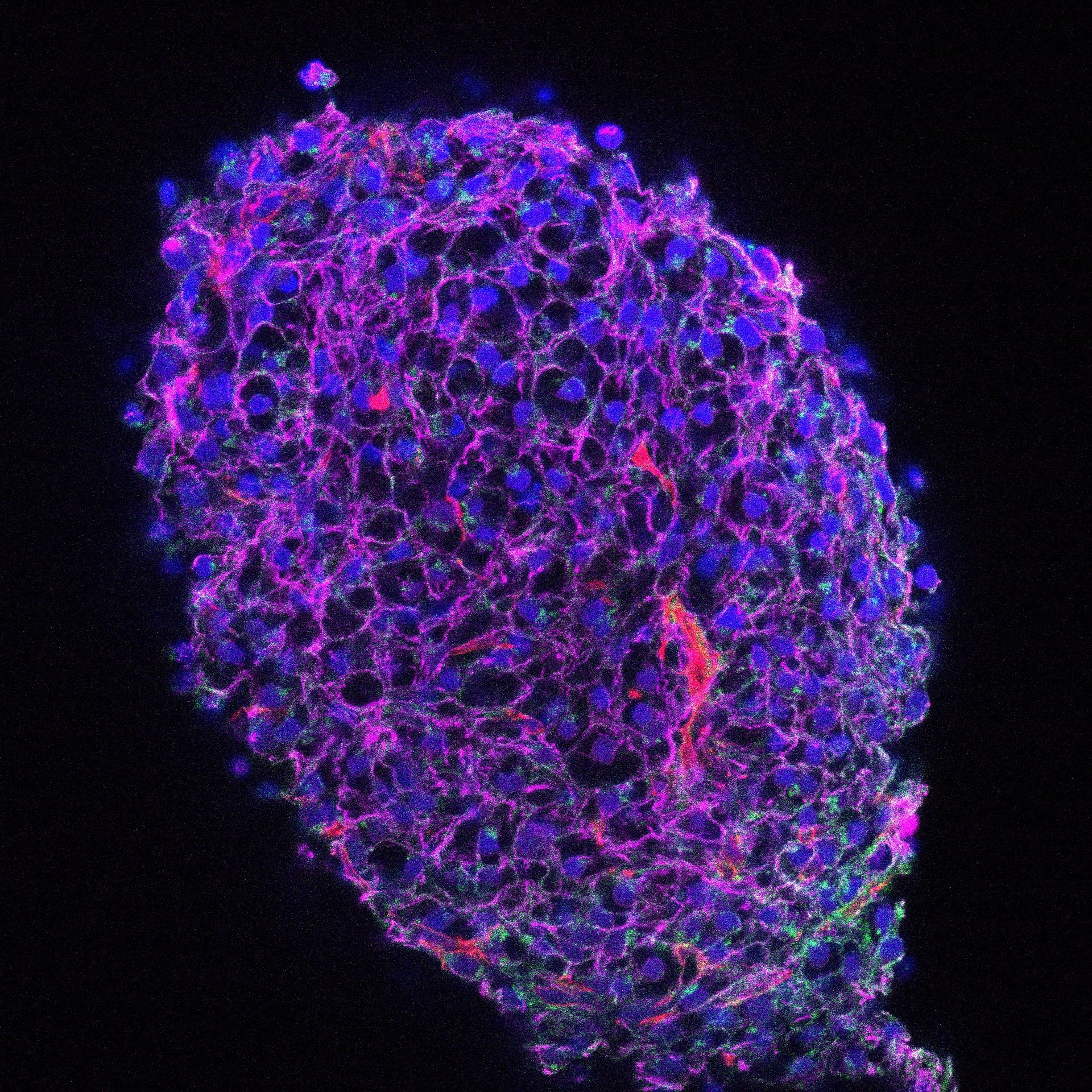
Advanced Organoids
We use EPSCs to generate organoids that replicate human tissue, driving innovation in translational research.
Broad Application
EPSCs drive advances in regenerative medicine, drug discovery and cell therapy.
Unlocking EPSCs Capability
EPSCs closely resemble early human embryos at the 8-cell to morula stage and possess an extraordinary ability to generate both embryonic and extraembryonic lineages, including the placenta. This feature distinguishes EPSCs from other pluripotent stem cells.
With exceptional genetic and epigenetic stability, EPSC provides a reliable foundation for sustained research and therapeutic innovation. Their compatibility with precise genome editing technologies enables accelerated progress in studies of gene function and the development of targeted therapies.
How EPSCs help
From the Lab to Real World Use
EPSCs can be derived from embryos or reprogrammed from somatic cells. We have established EPSCs in multiple species, including human, mouse, porcine, and bovine.
This next-generation stem cell technology continues to drive transformative advances in immunology, infectious disease modelling, regenerative medicine, and drug discovery, representing a significant step forward in translating stem cell science into real-world medical solutions.
Copyright ©2026 All rights reserved | InnoHK Centre for Translational Stem Cell Biology